
From the Simpsons to the quest for Ulysses, there's only one step... Transdisciplinary illustration by two uninhibited teachers.
Publish at November 24 2025 Updated December 10 2025

In winter, we've all seen the sweep of the spreaders that come to de-ice our roads so we can drive without ice skates. But do you know how this phenomenon works? In your opinion, does the temperature of the ice rise or fall? How can this phenomenon be used to replace the refrigeration systems that are partly responsible for global warming? Let's take a closer look!
The salt we use in our kitchens is sodium chloride (NaCl). This is the same salt used for roads, although calcium chloride (CaCl) or magnesium chloride is sometimes used. On contact with ice, sodium chloride begins to dissolve in water, breaking down into chlorine (Cl) and sodium (Na) ions. This double phase change (solid salt "liquefies" and ice becomes liquid) creates a new mixture. And this mixture has a different freezing point, in this case -22°C instead of 0°C for pure water, and -7°C for water containing 1% salts.
The
mixture can therefore remain liquid at the surface, down to very low
temperatures. When we see this phenomenon, we might think that it's the
ice that has "warmed up". Well, it's not! To dissociate and pull water
molecules from the ice, the salt molecules need energy, which they find
by taking it from the ice crystals. And when solid water gives up its
energy, its temperature drops sharply; this process is said to be
endothermic. The phenomenon therefore had the effect of cooling the
system, not heating it (1).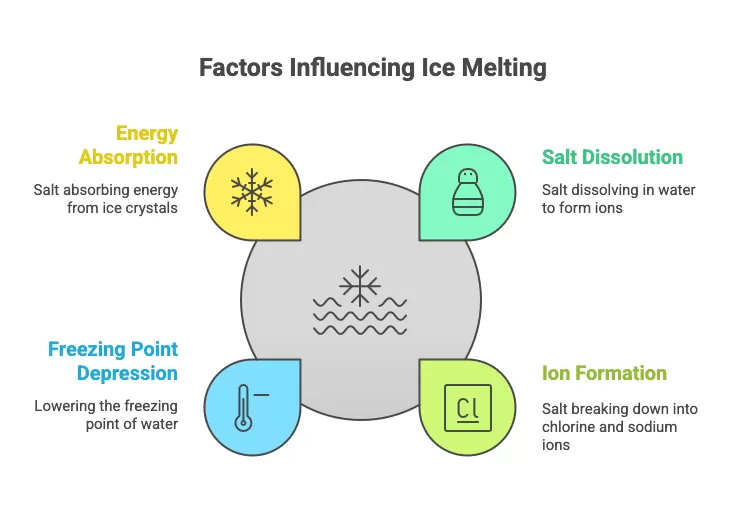
Scientists at the Lawrence Berkeley National Laboratory and the University of California, Berkeley, have used this idea as the basis for their work on environmentally-friendly refrigeration systems. In fact, the systems used to create cold evacuate heat by means of a fluid that absorbs excess temperature as it expands. You only have to touch a refrigerator compressor to realize that it's heating your kitchen!
However, the systems used in refrigeration processes rely on gases such as hydrofluorocarbons, which produce a very high greenhouse effect (2) and are regularly banned by regulations for new equipment (3).
Experiments were carried out in 2022 (4) by Berkeley teams to melt ethylene carbonate with a salt of iodine and sodium by applying a weak electromagnetic field. This common organic solvent, used in lithium-ion batteries, is produced from carbon dioxide (CO2). The ionocaloric cycle then follows four stages;
Ionocaloric cycle
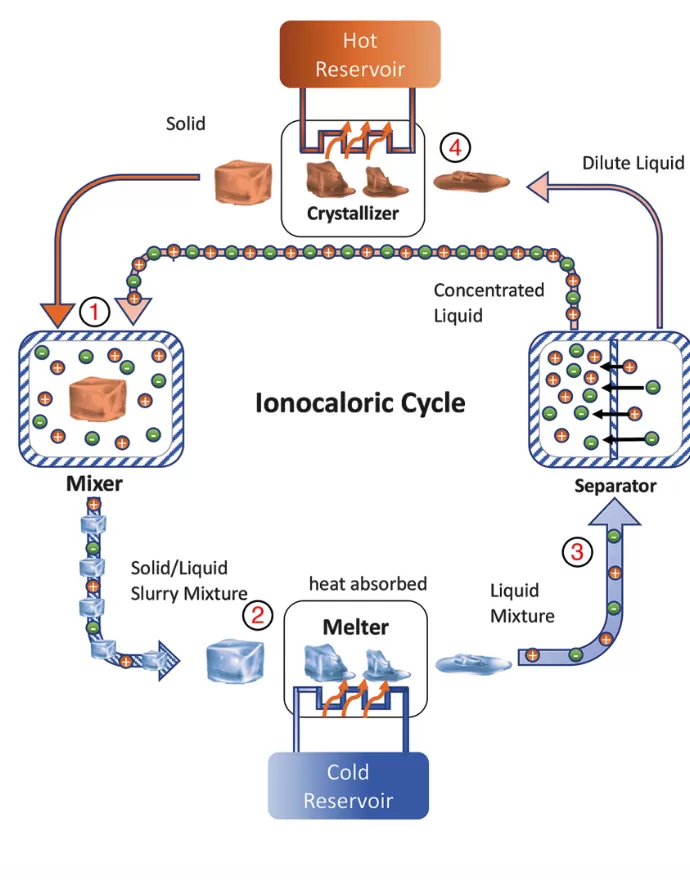
Source: Lilley & Prasher - Revue Sciences - 22/12/2022- (4)
This process could therefore have a negative impact on the climate, with the environment cooling down instead of warming up. On the other hand, the CO2 required to produce the solvent would make it possible to reduce emissions of this greenhouse gas, by reusing CO2 produced by other industrial processes, even if this solution is not viable in the long term (5).
The so-called ionocaloric effect is also reversible. Removing ions from a system can heat it up. This phenomenon could therefore also be used to replace heating systems, in an environmentally-friendly way. Initial tests are encouraging.
"We have demonstrated how the ionocaloric effect can lead to performance superior to that of other caloric materials. Even higher coefficients of performance, temperature rises and power density are possible with further improvements."
Progress remains to be made, therefore, in experimenting with more complex fluid systems for greater efficiency, and in producing the membranes needed for more efficient ionic separation, but the future of refrigeration may already be within test tube range.
Illustration: ShutterStock - 2460978767
References:
1- "Let it cool, with a pinch of salt"- Emmanuel Defay- Revue Science- December 2022- https://www.science.org/doi/10.1126/science.adf5114
2- "R410A refrigerant: characteristics, bans, replacement fluids..." 10/6/2025- https://www.dalkiafroidsolutions.com/fluide-frigorigene-r410a-caracteristiques-interdictions-fluides-de-remplacement
3- "La résillience climatique... par la prévention et le respect des lois" Virginie Guignard Legros - Thot Cursus- 17/1/2024 https://cursus.edu/fr/29763/la-resillience-climatique-par-la-prevention-et-le-respect-des-lois
4- "Cycle de réfrigération ionocalorique" Lilley & Prasher - Revue Sciences - 22/12/2022- https://www.science.org/doi/10.1126/science.ade1696
5- Is carbon capture a real solution - Alexandre Roberge - Thot Cursus - 5/10/2022- https://cursus.edu/fr/25362/capter-le-carbone-est-il-une-veritable-solution